Bcs Classification System
- Bcs Classification System
- Biopharmaceutical Classification System (bcs) Class Ii
- Bcs Biopharmaceutical Classification System
Llama firearms serial numbers. The Biopharmaceutics Classification System (BCS) is a scientific framework for classifying drug substances based on their aqueous solubility and intestinal permeability. Image pro plus 7.0. This classification system was devised by Amidon et al.[1] This concept underlying the BCS published finally led to introducing the possibility of waiving in vivo. Bcs classification system 1. BIOPHARMACEUTICS CLASSIFICATION SYSTEM 2. Contents• Introduction• Overview of the Classification system• Applications• Conclusion• References 3. The Biopharmaceutics Classification System is a system to differentiate the drugs on the basis of their solubility and permeability. This system restricts the prediction using the parameters solubility and intestinal permeability.
Five Nights at Freddy's Review Fear-starved gamers For a neat twist on the survival horror genre, fear-starved gamers should definitely consider checking out Five Nights at Freddy's, a recent indie release which has seemingly taken the gaming world by storm and which has already spawned a sequel. Fnaf 2 free download full game. This game requires download and install on local hard drive to play. Nightmarish story The story is a nightmarish take on the movie Night at the Museum and places players in the shoes of Mike Schmidt, a security guard at a pizza joint which just happens to be under threat by the store's malfunctioning animatronic animal mascots, including Freddy Fazbear, Chica the Chicken and Foxy the Pirate Fox. The game is a fiendishly clever one, with a deceptively simple concept at its core and which is backed up by some wonderful design work that together create a highly memorable experience.
Bcs Classification System
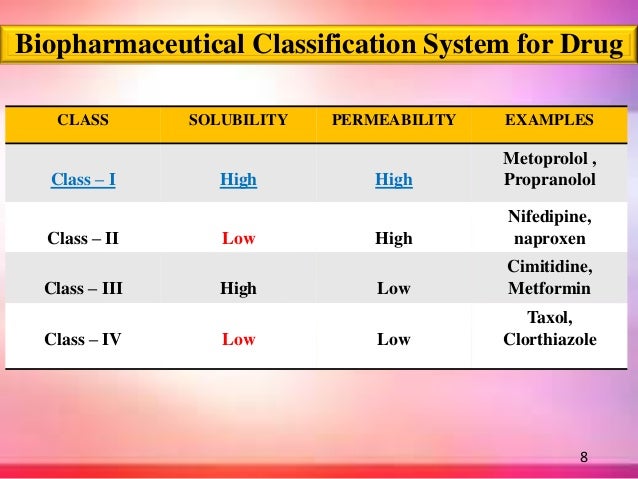

Biopharmaceutical Classification System (BCS) is a scientific framework for classifying drug substances based on their aqueous solubility and intestinal permeability
when the dissolution rate is much greater than the gastric emptying, dissolution is not likely to be rate-limiting
the pharmaceutical industry

Biopharmaceutical Classification System (bcs) Class Ii
The position of rifampicin with respect to the biopharmaceutics classification system (BCS) was explored on the basis of its aqueous solubility and intestinal permeability. The aqueous solubility was determined between pH 1 and 7 by the conventional shake-flask method. Permeability coefficients of rifampicin and the US FDA listed low (furosemide, ranitidine) and high permeable (caffeine, naproxen) model drugs were determined in the three segments of the rat intestine by employing the everted gut sac model. The samples were analyzed by HPLC. The maximum human single dose of rifampicin (600 mg) was insoluble in 250 ml of aqueous media between pH 3 and 7. The determined apparent permeability coefficient (Papp) values for rifampicin were 4.856×10−6, 2.117×10−6, and 2.149×10−6 cm/sec in the rat duodenum, jejunum and ileum, respectively. These values were similar to those of the low permeable drugs, ranitidine and furosemide, for which Papp values were determined to be 1.767×10−6−2.426×10−6 and 2.469×10−6−3.008×10−6 cm/sec, respectively. The determined values for high permeable model drugs, viz., caffeine and naproxen, were ∼10–20 folds higher than rifampicin or even ranitidine and furosemide. The study suggests that rifampicin is a drug with low solubility and even low intestinal permeability and hence qualifies to be classified in BCS Class IV, instead of Class II where it is being categorized presently. The contention is supported by the reported data on the permeability of the drug through cell monolayers, the mass balance and the absolute bioavailability values in the literature, and the data for rifampicin according to “Lipinski's rule of five”.